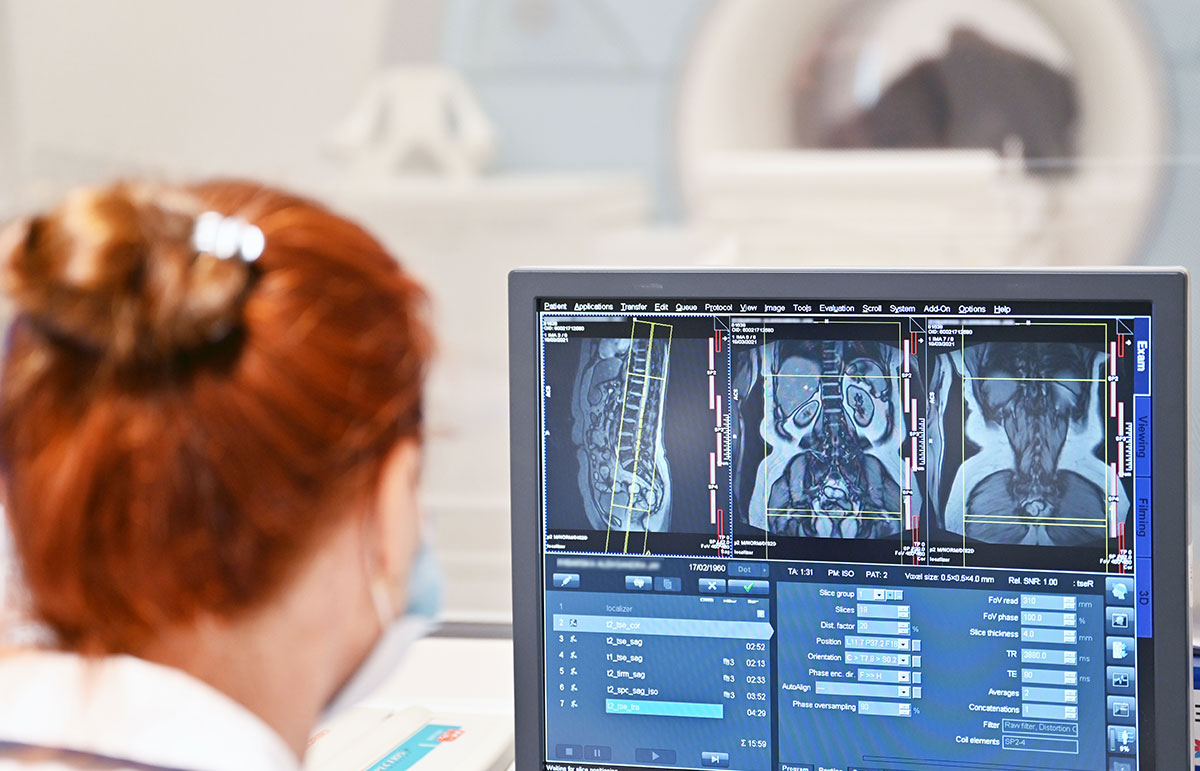
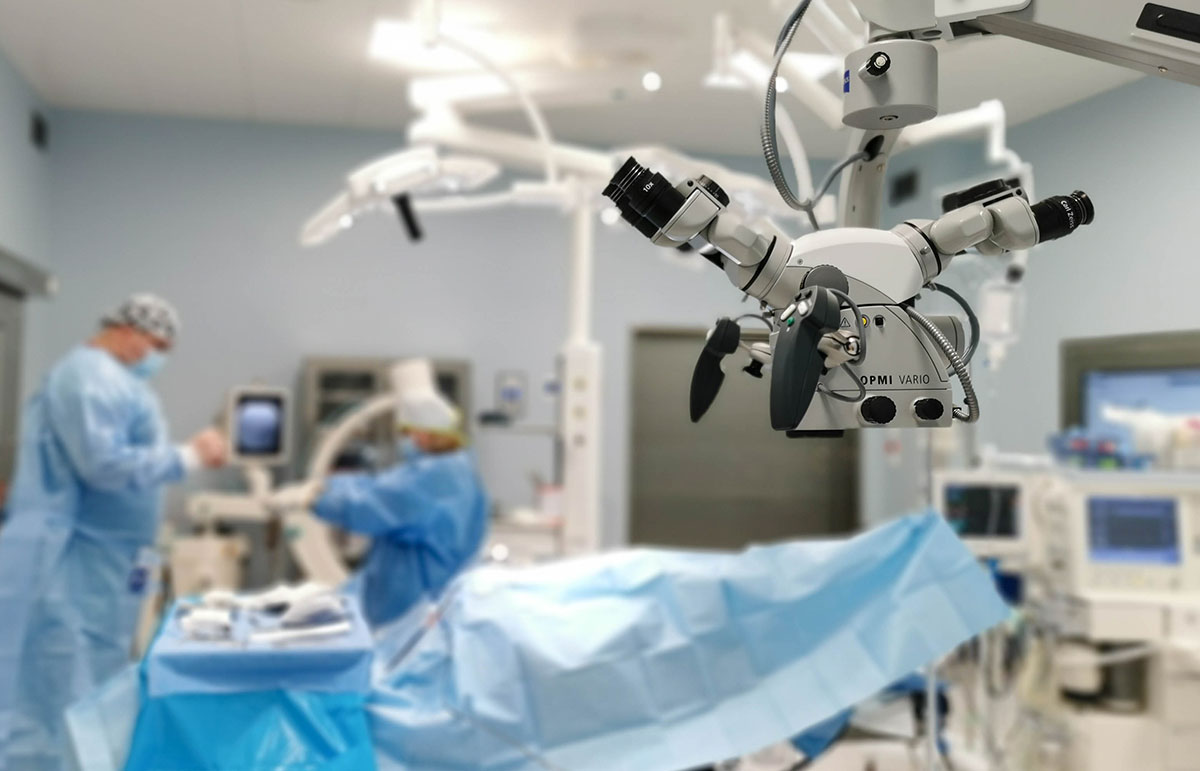
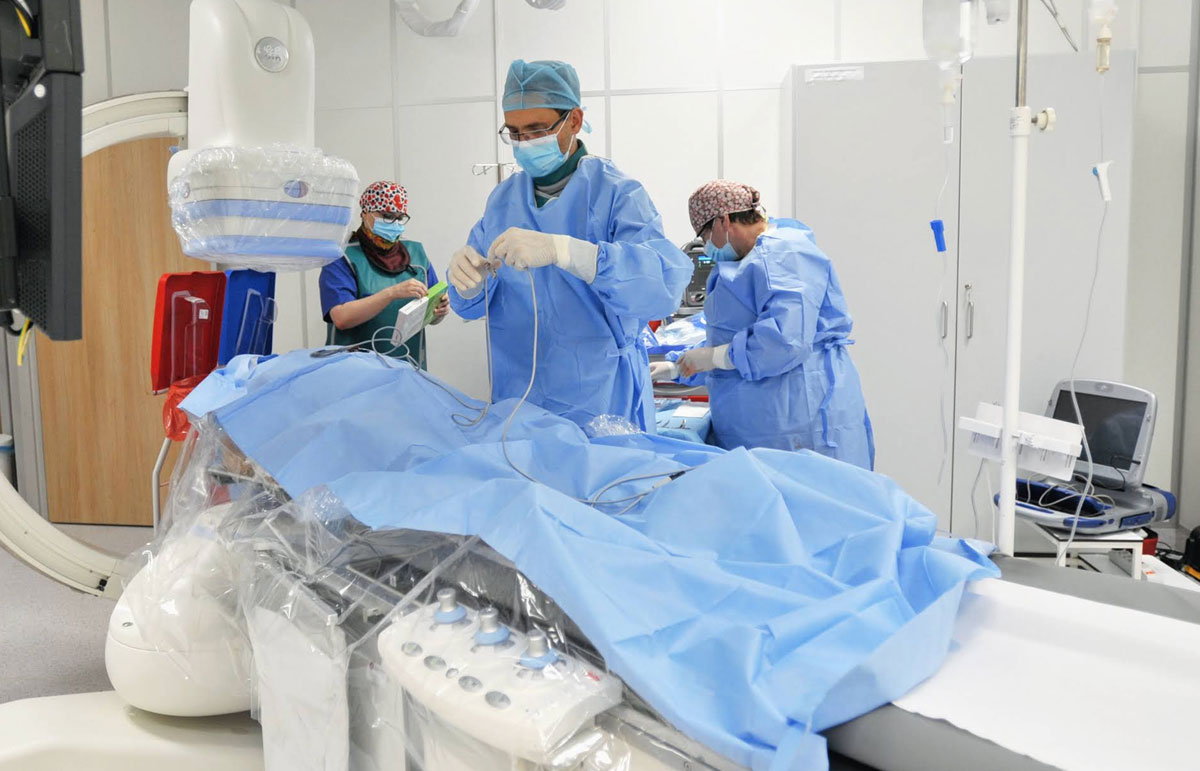
Clinical Trials Department was launched in 2021. Medical innovations emerge every day in different parts of the world. Within the Clinical Trials Department, we cooperate with International Clinical Research Organizations and Pharmaceutical Companies that are determined to implement innovative solutions, especially in the area of cardiology, invasive cardiology, gynaecology, general surgery and internal medicine.
Clinical trials at Żywiec Hospital are carried out in accordance with legal standards in force in Poland, international regulations and Good Clinical Practice guidelines, recognized as the gold standard in conducting clinical trials.
The research is carried out using modern equipment by a qualified team, experienced in the field of cardiology, including cath lab procedures. This translates into the safety of participating patients and the high quality of data collected in the clinical trials conducted in accordance with the ALCOA-C principles.